Leukocyte adhesion deficiency (LAD) is an immunodeficiency disorder associated with recurrent infections. This genetic disorder has been described in humans, cats, dogs and cattle. Human LAD is classified into three types (LAD-1 to -3), of which type 1 most closely resembles LAD in cats. Feline LAD is a very difficult condition and, if left untreated, it can be lethal for affected kittens.
Background information
Leukocytes, or white blood cells, are the cells of the immune system involved in the protection of an organism from diseases or other strange invaders. Leukocytes are classified into five main types: neutrophils, eosinophils, basophils, lymphocytes and monocytes (LaFleur-Brook, 2008).
Neutrophils are phagocytes, involved in the innate immune system. Early in an inflammation (i.e. due to a bacterial infection), neutrophils are the first cells to respond and migrate to the site of inflammation. They migrate through the blood vessels and interstitial tissues, and within minutes they are recruited to the affected site (this migration of neutrophils to the site is called chemotaxis).
When at the site, neutrophils engage in three main strategies of destroying the invaders: phagocytosis (ingestion of the foreign molecule), degranulation (production of proteins with antimicrobial properties) and generation of neutrophil extracellular traps. In some cases, neutrophils can't combat the infection themselves, and other immune cells are recruited to the site, too (Yoo et al, 2011 & Barer, 2012).
An important protein crucial for successful adhesion and cell surface signaling of neutrophils is CD18. This protein, among other functions, enables the migration of neutrophils out of the blood cells, their chemotaxis, immune cell interactions, immune recognition, and cell-killing. Cats affected with LAD1 suffer from a deficiency of the β subunit of CD18.
This deficiency among other effects, results in poorly-functioning neutrophils, which is what ultimately leads to recurrent infections (Amin Aanout, 1990).

Genetics
The deficiency of leukocyte integrin CD18-β subunit is caused by an autosomal recessive mutation in the integrin β-2 subunit (ITBG2) gene. This subunit of the CD18 protein forms a heterodimer (a complex composed of two different proteins) with one of 4 different α-subunits of the CD11 protein. Together, CD11 and CD18 form CD11/CD18 integrins, which are expressed on all leukocytes’ surface membranes.
They enable the normal functioning of neutrophils and are involved in the activation of T-lymphocyte proliferation (Bauer et al, 2017). The mutation is inherited in an autosomal recessive mode, meaning that a cat must inherit two mutated copies of the gene (alleles), one from each parent, in order to be affected by the disorder. Cats who carry only one mutated allele and one healthy allele remain silent carriers (they can pass on the trait to the offspring but aren't affected themselves). If two silent carriers cross, there is a 25% chance that each kitten will be affected by LAD. Two affected cats give only affected offspring.
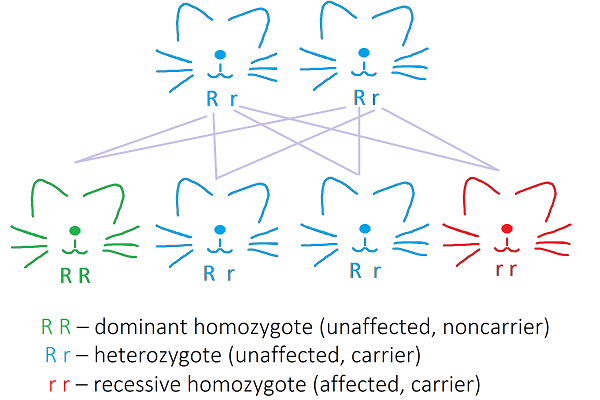
Autosomal recessive mode of inheritence
Symptoms and diagnosis
The clinical presentation of LAD in cats involves persistent leukocytosis (high white blood cell count), chronic bacterial infections, poor responsiveness to antibiotics, impaired wound healing, gastroenteritis, anterior uveitis, subcutaneous abscesses, and epidermal sloughing/cellulites with sepsis. In some cases, progressive juvenile gingivitis and periodontitis leading to early loss of dentition and the ascending umbilical infection have been described too. The diagnosis of LAD can be successfully done with flow cytometry (Bauer et al, 2017).
Treatment and prognosis
LAD is a serious disorder, and if left untreated, affected kittens rarely reach adulthood. Intensive long-term antimicrobial therapies and, in some cases, bone marrow transplantation have been found to be successful (Bauer et al, 2017). Note: It is not recommended to breed the carriers of the mutation in order to prevent the progression of the condition to offspring.

Like many other immunodeficiency disorders, leukocyte adhesion deficiency is a very challenging and detrimental genetic disease. Luckily, it is possible to successfully treat and control LAD in cats as long as the diagnosis is accomplished correctly and in a timely manner.
Bibliography 1. Amin Aanout, M. (1990). Structure and Function of the Leukocyte Adhesion Molecules CD11/CD18. Blood 75 (5): 1037–1050. 2. Barer, M.R. (2012). The natural history of infection. Medical Microbiology. Elsevier. doi:10.1016/b978-0-7020-4089-4.00029-9 3. Bauer TR, Pratt SM, Palena CM, Raj K, Giger U (2017). Feline leukocyte adhesion (CD18) deficiency caused by a deletion in the integrin beta-2 (ITGB2) gene. Vet Clin Pathol 46(3): 391-400. doi: 10.1111/vcp.12526 4. LaFleur-Brooks M (2008). Exploring Medical Language: A Student-Directed Approach (7th ed.). St. Louis, Missouri, US: Mosby Elsevier. p. 398. ISBN 978-0-323-04950-4. 5. Yoo, SK; Starnes, TW; Deng, Q; Huttenlocher, A (2011). Lyn is a redox sensor that mediates leukocyte wound attraction in vivo. Nature 480 (7375): 109–12. doi:10.1038/nature10632
