Gangliosidosis is a group of lipid storage disorders characterized by the accumulation of lipids – gangliosides in neurons. The disease was identified both in humans and in cats. Similarly to humans, several types of gangliosidosis in cats have been described.
Human gangliosidosis is classified into two types, GM1 and GM2. The second type is further classified into three subtypes: GM2A (Tay-Sachs disease), GM2AB (AB variant) and GM2B (Sandhoff disease or 0 variant). All of the variants of the human disease have been identified in cats except for the Tay-Sachs (GM2A).
Background information
Cats suffering from gangliosidosis have a disrupted metabolism of certain lipids (gangliosides). This results in the lipid accumulation within the cells in the body (particularly neurons in the central and peripheral nervous system). This eventually leads to progressive destruction of the nerve cells in the brain and spinal cord. Two types of gangliosidoses have been described in both cats and humans: type 1 and type 2.
Type 1 (GM1 gangliosidosis) is characterized by a deficiency of an enzyme called beta-galactosidase. Type 2 is a group of several related conditions characterized by a deficiency in the beta-hexosaminidase enzymes (A and/or B).
In humans type 2 (GM2) gangliosidoses are classified in three subtypes: GM2A (Tay-Sachs disease), GM2AB (AB variant) and GM2B (Sandhoff disease or 0 variant). Tay-Sachs disease has not been described in cats yet, but AB variant and Sandhoff disease have. All three GM2 diseases represent a distinct molecular point of failure in the activation of the beta-hexosaminidase enzymes.

Genetics
GM1 gangliosidosis is caused by an autosomal recessive mutation in exon 14 of the GLB1 gene which codes for beta-galactosidase. Among other functions, this enzyme is responsible for the hydrolysis of lipids known as GM1 gangliosides. The mutation causes the deficiency of the respective enzyme which then leads to the accumulations GM1 gangliosides. This mutation has been identified in Siamese and Korat cat breeds.
GM2AB or AB variant of GM2 is caused by an autosomal recessive mutation in the GM2A gene. This gene codes for a protein called GM2 activator which is required for the normal functioning of the beta hexosaminidase A. The respective mutation disrupts the functioning of GM2 activator and the activation of beta-hexosaminidase A fails. Beta-hexosaminidase A breaks down the lipids called GM2 gangliosides. The failure of its activation leads to the accumulation of these lipids in the neurons.

GM2A and GM2B are gangliosidoses characterized by the deficiency in the beta hexosaminidases A and B. Beta-hexosaminidase consists of two subunits: alpha (produced by HEXA gene) and beta (produced by HEXB gene).
GM2A is caused by a mutation in the HEXA gene and therefore results in the deficiency of the beta-hexosaminidase A. This disease is more commonly known as Tay-Sachs disease and has not been described in cats yet.
GM2B is a result of an autosomal recessive mutation in the HEXB gene which leads to the deficiency in both beta hexosaminidases A and B. This mutation causes a deficiency of the respective enzymes which then leads to the accumulations of lipids known as GM-2 gangliosides. This mutation has been identified in Burmese and Korat cat breeds and is famously known as the Sandhoff disease or the variant 0 of the GM2.
Only homozygotes are affected because all three genetic mutations leading to the three variants of gangliosidosis in cats are inherited by a recessive mode of inheritance.. Heterozygotes are silent carriers. If two heterozygotes are bred from, there is a 25% chance for each of kitten to be affected.
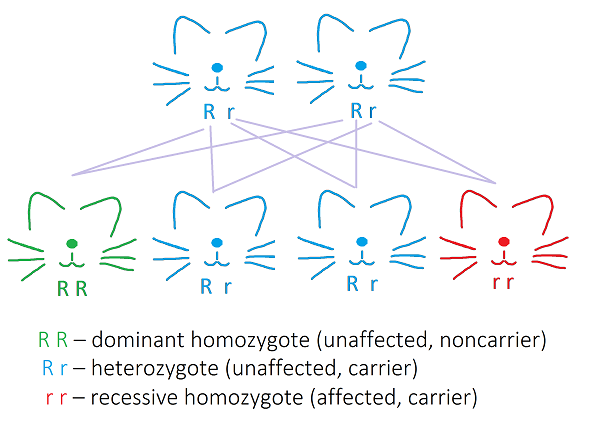
Recessive mode of inheritance
Symptoms and diagnosis
Affected cats typically develop progressive neurological signs such as ataxia, dysmetria, tremor and nystagmus (involuntary movement of the eyes). The clinical presentation of feline gangliosidosis may also include an enlarged liver, facial dysmorphism, visual impairment, clouded cornea and the presences of granules in the cytoplasm of leukocytes.
Today DNA tests are available for detection of all three known forms of feline gangliosidosis (GM1, GM2AB and GM2B). The DNA tests can reveal both silent carriers and affected cats (recessive homozygotes).
Treatment and prognosis
Unfortunately, this disease cannot be treated. Clinical presentation usually occurs between 1 and 5 months of age and affected kittens typically die around the age of 8-10 months. The only preventative method is avoidance of breeding from silent carriers. This would help reduce the incidence of the mutation in the feline population. This disease is included in the Basepaws Health Report.

Gangliosidosis is a lethal genetic disease in cats. While the adequate treatment hasn't been identified yet, we now know the genetic cause and background of the disease which allows us to prevent the breeding of silent carriers and succession of the disease through the generations.
Hopefully, the advancement of medical engineering and gene therapy might one day allow us to develop an adequate treatment of this and similar genetic diseases too.
Bibliography
1. Bradbury AM, Morrison NE, Hwang M, Cox NR, Baker HJ, Martin DR (2009). Neurodegenerative lysosomal storage disease in European Burmese cats with hexosaminidase beta-subunit deficiency. Mol Genet. Metab. 97(1):53-9. doi: 10.1016/j.ymgme.2009.01.003
2. Martin DR, Rigat BA, & Foureman P. (2008) Molecular consequences of the pathogenic mutation in feline GM1 gangliosidosis. Molecular Genetics and Metabolism 94: 212-221.
3. Muldoon LL, Neuwelt EA , Pagel MA, & Weiss DL. (1994) Characterization of the molecular defect in a feline model for type II GM2-gangliosidosis (Sandhoff Disease). American Journal of Pathology 144:1109-1118.
4. Neuwelt EA, Johnson WG, Blank NK, Pagel MA, Maslen-McClure C, McClure MJ, Wu PM (1985). Characterization of a new model of GM2-gangliosidosis (Sandhoff's disease) in Korat cats. J Clin Invest. 76(2): 482–490. doi: 10.1172/JCI111997
