Cystinuria in cats is an inherited metabolic disease that is relatively common in dogs, but rare in cats (Mizukami, 2016). The condition is characterized by defective amino acid reabsorption, leading to the formation of cystine stones in the kidney, ureter and the bladder (cystine urolithiasis). This can lead to urinary obstruction. In cats, only two cystinuria types have been identified so far: I-A and II-B (Mizukami et al, 2015 & Mizukami et al, 2016).
Background information
Cystinuria is a congenital disease characterized by high cysteine concentrations in the urine. This can lead to the formation of cystine uroliths (kidney, bladder and urethra stones) which can further cause a urinary obstruction. Besides cats, this disease has also been described in humans and dogs.
In humans, cystinuria is classified into four types: I-A and I-B (autosomal recessive cases) and II-A and II-B (autosomal dominant cases). Canine cystinuria is classified in three human-like types: I-A, II-A and II-B, and a canine-restricted type III. This classification is based on the underlying causes leading to the clinical presentation. In cats, only two cystinuria types have been identified so far: I-A and II-B (Mizukami et al, 2015 & Mizukami et al, 2016).

Cystinuria in cats
The kidneys are the two bean-shaped organs of the renal system which are involved in the filtration of the blood. Kidneys process the blood through filtration, reabsorption, secretions and excretion. In the first step, the blood is filtered. Cells, proteins and other large molecules are retained, while smaller molecules are filtered into an ultrafiltrate that will eventually become urine. Then, water (~55%) and electrolytes are reabsorbed from the ultrafiltrate back into the blood system and terminal metabolic products (waste products) are secreted from the blood into the ultrafiltrate. Finally, ultrafiltrate is excreted into the ureters, where it becomes urine. Through this process of filtration and excretion, kidneys help maintain homeostasis in the kitty's body. In cystinuria, however, the amino acid cysteine is inadequately reabsorbed from the ultrafiltrate. This results in excessive cysteine concentrations in the urine and can lead to the formation of cystine uroliths.
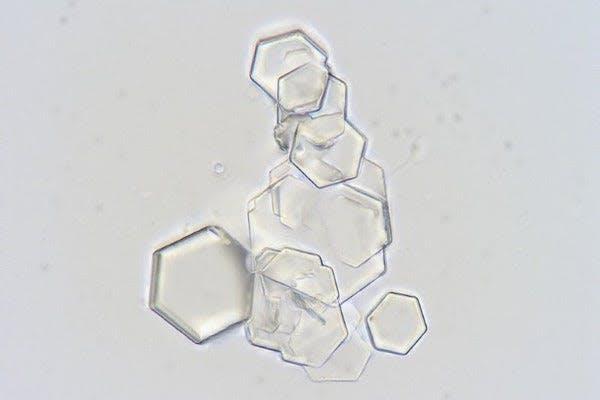
The cystine crystals found in the urine sediment of a male dog (source)
Genetics:
In humans and dogs, cystinuria is caused by a mutation in one of the two genes: SLC3A1 or SLC7A9. Mutations in SLC3A1 gene lead to either I-A cystinuria (caused by a recessive mutation) or II-A cystinuria (caused by a dominant mutation). I-B cystinuria (restricted to humans) and II-B cystinuria are caused by recessive and dominant mutations in the SLC7A9 gene (respectively).
In cats, two mutations associated with cystinuria have been identified. The first is an autosomal recessive mutation in the SLC3A1 gene, and the second is the autosomal dominant mutation in the SLC7A9 gene. The first mutation leads to a feline cystinuria case which highly resembles the canine type I-A cystinuria. The second mutation leads to feline II-B cystinuria (Mizukami et al, 2015 & 2016).
Feline I-A cystinuria is caused by a missense mutation in the SLC3A1 gene which codes for a globular protein rBAT. The mutation results in a deleterious amino acid substitution of a conserved arginine residue in the rBAT protein. The rBAT protein is responsible for cystine uptake and the described abnormality disrupts its function.
Aside from being more commonly seen in dogs, this particular mutation has previously been described in humans as well. (Mizukami et al, 2015 & 2016). It is inherited in an autosomal recessive manner (Figure 1), which means that only recessive homozygotes will be affected. Heterozygotes will remain silent carriers, and although they will remain unaffected, they can still pass the defect on to the next generation.
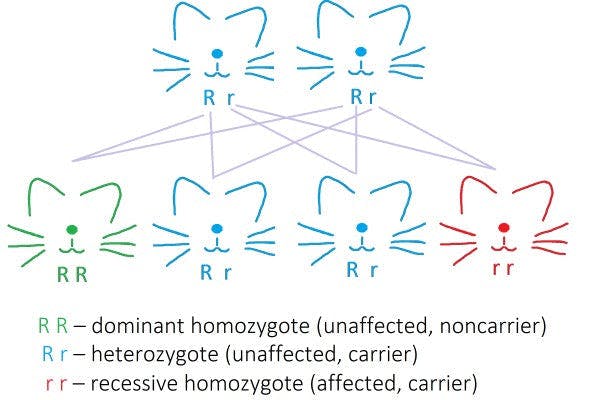
Figure 1: Autosomal recessive mode of inheritance
Feline II-B cystinuria, on the other hand, is associated with a few variants of the SLC7A9 gene which codes for an intramembrane transporter protein called b0,+AT. This protein is involved in the transport of cystine and neutral and dibasic amino acids. A few variants of the SLC7A9 gene have been found in cats which result in a modified version of this protein which has disrupted functions.
The respective genetic variants result in the substitution of one of the 3 highly conserved amino acid residues in this protein (valine to glutamic acid, threonine to methionine or aspartic acid to asparagine) (Mizukami et al, 2016). These mutations are dominant and will affect all carriers (Figure 2).
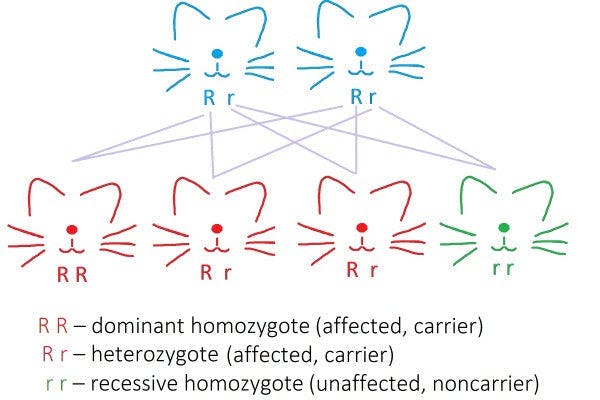
Figure 2: Autosomal dominant mode of inheritance
Symptoms and diagnosis:
Cats suffering from cystinuria of any type will have elevated levels of cysteine in their urine and can develop uroliths in the kidneys, bladder and urethra (Mizukami et al, 2015 & Mizukami et al, 2016). This can also lead to blockages in the urinary tract. Urethral obstruction is a serious medical emergency which requires immediate veterinary attention.
This is because the obstruction of urethra can lead to renal failure and uremia. If left untreated, such conditions may lead to death in less than 72 hours. Patients diagnosed with this disease may also experience pain when urinating and overall distress. Remember that in cases of urethral obstruction, there will be little or no urine passed! For the diagnosis of cystinuria, urinalysis is the most common approach in feline diagnostics. The urinalysis is based on the detection of cysteine in the urine. However, a negative result of this test is not reliable because it does not guarantee that the cat doesn't suffer from this disease.
Treatment and prognosis:
Cystinuria is a genetic defect that, unfortunately, cannot be treated. However, the prognosis can vary. Cats (and dogs) suffering from cystinuria don't always form uroliths and can live long and healthy lives before (if any) clinical presentation. It is not known why some cats are urolith-formers while others are not, but it is possible that diet and other environmental factors influence the prognosis of the condition.

Cystinuria in cats
Cystinuria is a serious genetic defect with a variable prognosis. As in the case of most genetic diseases in cats, the best preventative method is detection of mutation carriers and their isolation from the breeding pool. Feline science is now developing more successful and thorough genetic tests for cats, like the Basepaws CatKit, which will hopefully be able to screen for numerous genetic diseases, enabling better feline healthcare.
Related articles:
Bibliography:
1. Mizukami K, Raj K, Giger U (2015). Feline Cystinuria Caused by a Missense Mutation in the SLC3A1 Gene. J Vet Intern Med 29(1): 120-125. doi: 10.1111/jvim.12501
2. Mizukami K, Raj K, Osborne C, Giger U (2016). Feline Cystinuria Associated with Different SLC7A9 Gene Variants in the Cat. PLoS One 11(7). doi: 10.1371/journal.pone.0159247
