Autoimmune lymphoproliferative syndrome (abbr. ALPS) is a lymphoproliferative disorder (LPD) distinguished by massive enlargement of lymphatic nodes and spleen caused by the accumulation of lymphocytes. The disease is caused by irregular lymphocyte apoptosis.
This is a lethal genetic condition that has, for now, only been identified in British Shorthair cats. Kittens suffering from ALPS show signs around the age of 6-8 weeks and die or are euthanized shortly after (Aberdein et al, 2017). The disease shows similarities to the human disorder autoimmune lymphoproliferative syndrome (ALPS), also known as the Canale-Smith syndrome.
Background information
Lymphocytes are a type of white blood cells (Figure 1) which play an important role in the immune system. There are two types of lymphocytes: T cells and B cells. These cells are the main type of cells in lymph. Lymph is the fluid of the lymphatic system. It is similar to blood, but it is formed from the interstitial fluid (extracellular fluid) collected through lymph capillaries.
Aside from the circulating lymph, the lymphatic system also consists from the lymphatic organs (thymus, bone marrow, lymph nodes and spleen) and lymphatic vessels. In the autoimmune lymphoproliferative syndrome, the proliferation of lymphocytes is disrupted which results in their accumulation in the lymphatic system leading to severe health difficulties. This accumulation is a result of a disrupted cellular process called apoptosis.

Autoimmune lymphoproliferative syndrome in cats
Apoptosis is highly controlled, programmed cell death. Unlike necrosis, traumatic cell death, apoptosis is a regulated process which ensures the balance with the new cells arising from the stem cell populations and removes all the old, sick or dysfunctional cells from the body.
In ALSD, however, the apoptosis of lymphocytes is disrupted which alters their survival. This results in their accumulation in lymphatic nodes and spleen and leads to lymphadenopathy (enlargement of the lymph nodes) and splenomegaly (enlargement of the spleen) (Rao & Straus, 2006).
The disrupted apoptosis of lymphocytes in ALPS is a result of a defect in Fas-mediated apoptotic pathway. This pathway is a chain of reactions which lead to self-degradation of the cell and terminate with phagocytosis and removal of the cellular structures. Because of the importance of the regulatory role apoptosis has, the apoptotic processes are highly conserved across species. Apoptosis can be initiated internally or externally.
When the apoptosis is triggered externally, the caspase enzymes involved in apoptotic pathways are activated through death-receptor-ligand interactions at the cell surface. These receptors at the cell surface are members of the tumor necrosis factor (TNF) receptor family and contain a domain critical for the delivery of external apoptotic signals.
One of the most important receptors from this group is Fas receptor. Fas binds to Fas ligand (FasL) or to agonistic antibodies and triggers trimerization and recruitment to the Fas death domain of the Fas-associated death domain protein (FADD). This is followed by caspase activation and subsequent cell death.
Genetics
In humans, mutations across different locations in the Fas gene have been identified as the underlying cause of ALPS in three-quarters of patients (Rao & Straus 2006). In cats, a recessive mutation in the FASL gene has been identified (Aberdein et al, 2015 & Aberdein et al, 2017).
Cats carriers of two defect genes (recessive homozygotes rr) will suffer from ALPD, while cats with only one copy of the mutated gene (heterozygotes Rr) will be non-affected silent carriers. It is important NOT to breed heterozygotes as there is 25% of chance for each of the kittens to suffer from ALPD (Figure 2). By preventing the breeding of the carriers, we can prevent the succession of the disease to the offspring.
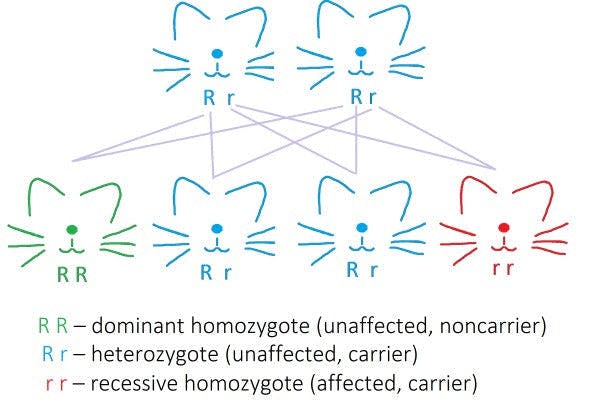
Figure 2: ALPS is caused by a recessive mutation in the FASL gene. The healthy copy of this gene is marked with R, and the defect copy with r. Every cat inherits two copies of all genes: one from each parent. Because the defect copy of the gene is recessive, it can only be expressed in those cats who carry two copies of the recessive gene (rr). These cats are recessive homozygotes. Cats with one copy of a healthy gene and one copy of a defect gene are heterozygotes (Rr) and remain unaffected, but can pass the defect on to the offspring. Finally, carriers of two healthy copies of the gene are dominant homozygotes (RR) and are unaffected non-carriers. If two heterozygotes are crossed there is a 25% chance for each kitten to inherit a defect copy of the FASL from each parent resulting in a homozygous genotype and the expression of the disease.
Symptoms and diagnosis
Symptoms of the disease start showing early, around the 6th week of age. Affected kittens rapidly develop lymphadenopathy, splenomegaly and regenerative hemolytic anemia. They show abdominal distension, lethargy and weight loss. Unfortunately, they shortly die or are euthanized after the clinical presentation (within 4 weeks) (Aberdein et al, 2017).
Treatment and prognosis
Unfortunately, the prognosis of the disease is not good. The kittens usually die shortly after the clinical presentation of the disorder (Aberdein et al, 2017). There are currently no known treatments for ALPD. The only preventative measurement is avoidance of breeding from the carriers. Genetic tests are now available for the detection of carriers which allows us to eliminate them from the breeding pool.

Autoimmune lymphoproliferative syndrome in cats
ALPS is a serious and lethal inherited feline condition. It is highly important not to breed silent carriers in order to avoid the progression of the disease in the feline population. By DNA testing your cat, you can make sure that your cat is not a silent carrier and is out of risk from passing the defect gene on. So far, this mutation has only been found in British Shorthair cats, but it is unknown if it's restricted to this and related breeds or not.
Bibliography
1. Aberdein D, Munday JS, Gandolfi B, Dittmer KE, Malik R, Garrick DJ, Lyons LA & 99 Lives Consortium (2017). A FAS-ligand variant associated with autoimmune lymphoproliferative syndrome in cats. Mammalian Genome 28, 47-55.
2. Aberdein D, Munday JS, Fairly RA, Vernau W & Thompson KG (2015). A novel and likely inherited lymphoproliferative disease in British Shorthair kittens. Veterinary Pathology 52, 1176-1182.
3. Rao V. K., Straus S. E. (2006). Autoimmune Lymphoproliferative Syndrome. Clinical Hematology. N. S. Young, S. L. Gerson and K. A. High. Philadelphia, Elsevier: 758-766.
